Bifunctional Approaches to Catalysis
Learn more about how we are leveraging metal-ligand cooperativity to carry out challenging catalytic reactions.
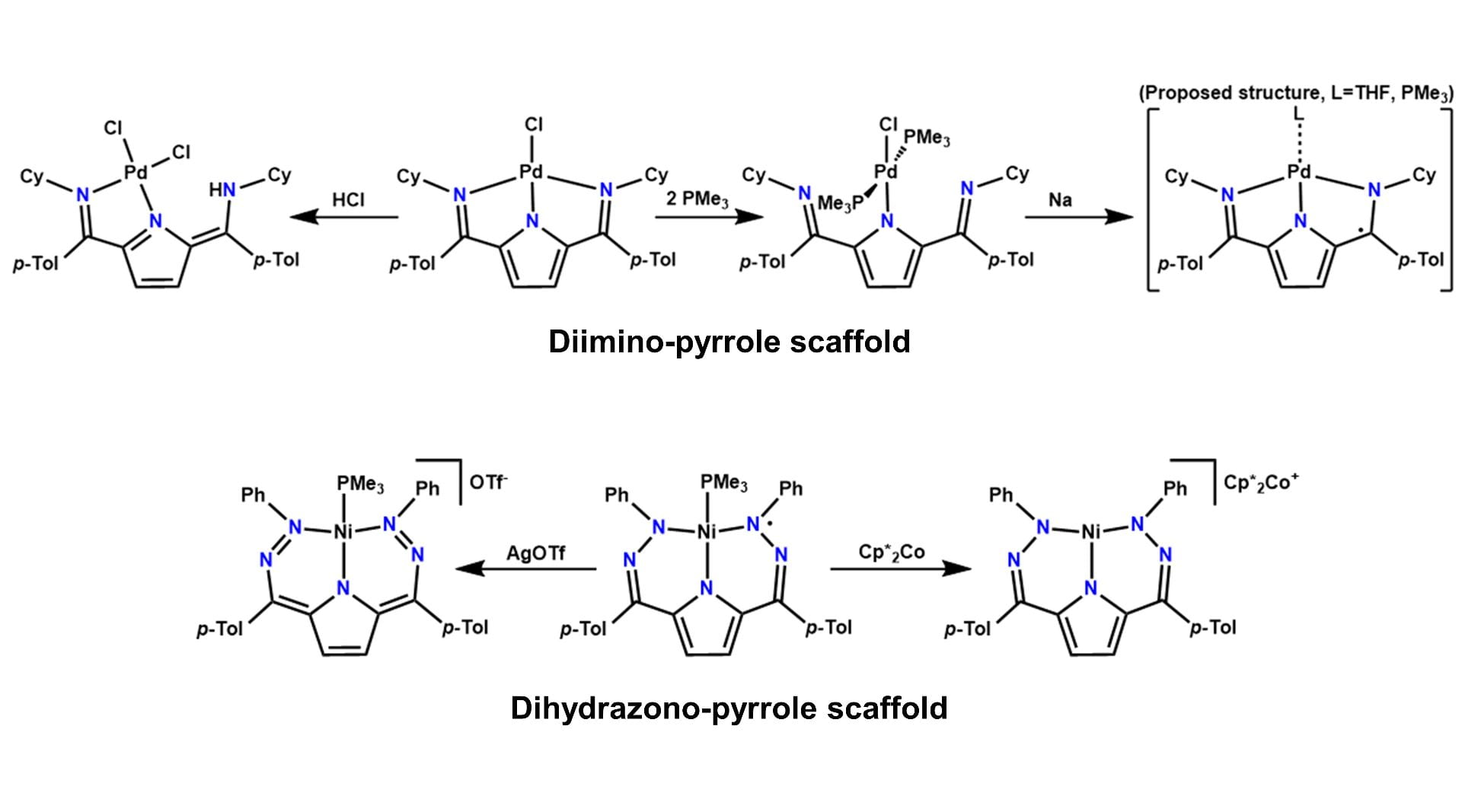
Variable Coordination, Redox, and Protonation States
Our approach to metal-ligand cooperativity uses a pyrrole-based scaffold with imines or hydrazones at the 2 and 5 positions of the pyrrole ring. In this scaffold, the nitrogens of the imine or hydrazone can function as pendant bases while the pyrrole ring can undergo a 2-electron redox couple. This allows for the net storage and transfer H-atoms or H2. Shown above are complexes that demonstrate these variable redox and protonation states.
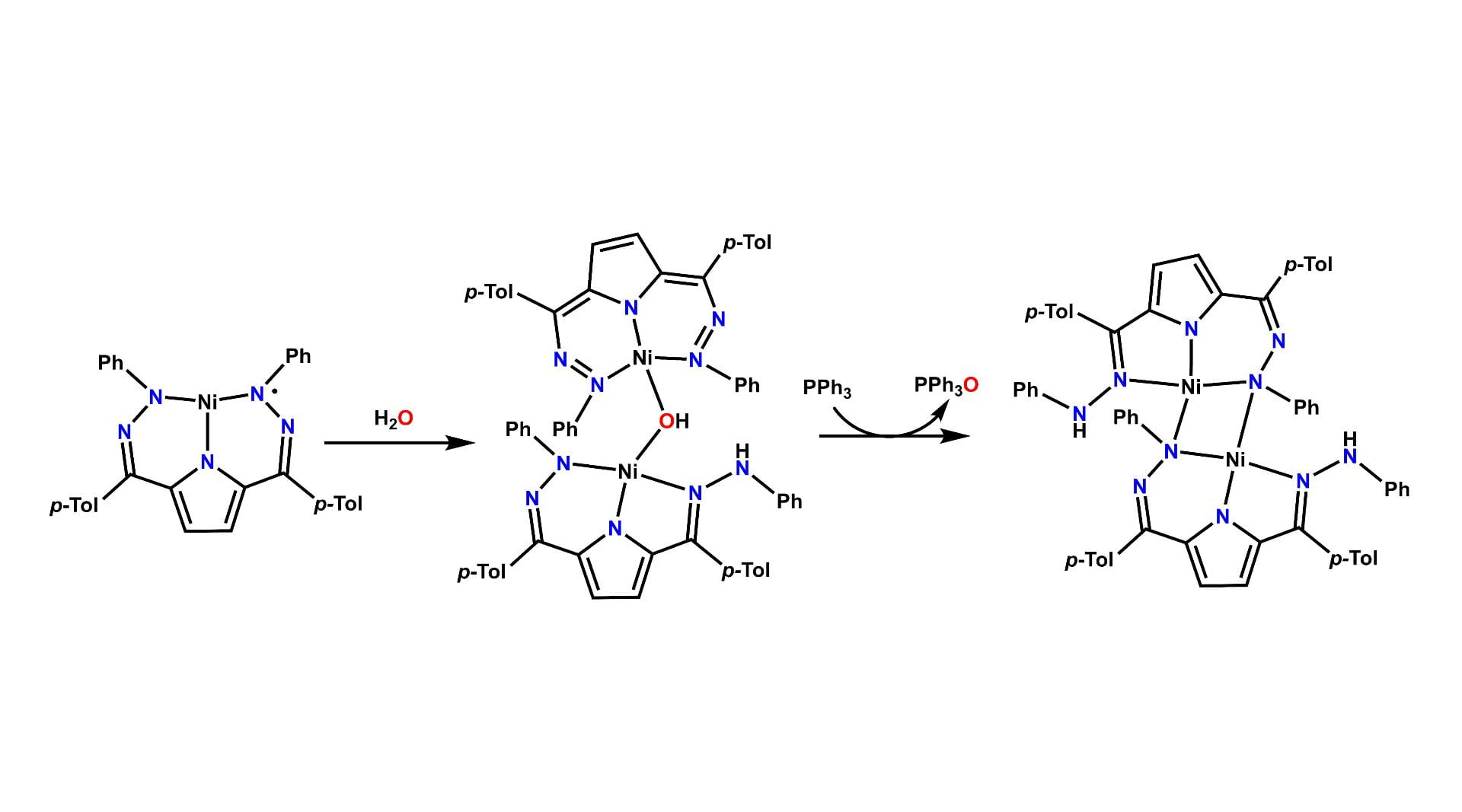
Homolytic Activation of Water
Metal-ligand cooperativity in a nickel complex utilizing the dihydrazono-pyrrole scaffold allows for the homolytic activation of an O-H bond in H2O. This demonstrates that novel and sometimes unexpected reactivity can be achieved via these bifunctional ligand systems. We are currently studying complexes of Fe, Co, Ni, and Cu bearing ligands with varying hydrazone substituents to discover what other rich chemistry this ligand system has in store.
Selected publications:
“Generation and Oxidative Reactivity of a Ni(II) Superoxo Complex via Ligand-Based Redox Non-Innocence” McNeece, A. J.; Jesse, K. A.; Xie, J.; Filatov, A. S.; Anderson, J. S. J. Am. Chem. Soc. 2020, ASAP. Read it!
“Nickel-Mediated Dehydrogenative Aryl-Aryl Homocoupling of a Bulky Phosphino-Pyridine” Mannava, V.; Jesse, K. A.; Anderson, J. S. Organometallics 2019, 38, 4554-4559. Read it!
“Neocuproine as a Redox-Active Ligand Platform on Iron and Cobalt” Jesse, K. A.; Filatov, A. S.; Xie, J.; Anderson, J. S. Inorg. Chem. 2019, 58, 9057-9066. Read it!
“Reversible Homolytic Activation of Water via Metal-Ligand Cooperativity in a T-shaped Ni(II) Complex” Chang, M-C; Jesse, K. A.; Filatov, A. S.; Anderson, J. S. Chem. Sci. 2019, 10, 1360-1367. Read it!
“Redox-Activity, Ligand Protonation, and Variable Coordination Modes of Diimino-Pyrrole Complexes of Palladium” McNeece, A. J.; Chang, M-C.; Filatov, A. S.; Anderson, J. S. Inorg. Chem., 2018, 57, 7044-7050. Read it!
“Ligand-Based Storage of Protons and Electrons in Dihydrozonopyrrole Complexes of Nickel” Chang, M-C.; McNeece, A. J.; Hill, E. A.; Filatov, A. S.; Anderson, J. S. Chem. Eur. J., 2018, 24, 8001-8008. Read it!
